-Life on Earth —
Our mission is to create a shared plan
for saving the planet’s most exceptional wild places.




Quality and Impact
High-quality genomic research relies on precise, cutting-edge techniques such as next-generation sequencing, CRISPR-based analysis, and high-throughput genotyping. These reliable methods ensure accurate identification of genes linked to stress tolerance, growth, and adaptation. By applying robust and reproducible genomic techniques, scientists can generate actionable insights to conserve biodiversity, improve crops, and restore ecosystems. Investing in advanced genomic methods strengthens our ability to protect and sustain Earth’s ecosystems effectively.
Technological Foundations of Functional Genomics
To fully exploit any model organism in functional genomics, two major categories of technologies must be established: physical genomics tools and biological genomics tools.
Physical genomics includes technologies such as genome sequencing, DNA mapping, and bioinformatics platforms. These tools are generally transferable between species and can be developed for virtually any organism, provided sufficient resources are available. They enable researchers to construct genome assemblies, identify genes, and analyze structural variations.
In contrast, biological genomics is highly species-dependent. It encompasses experimental approaches such as gene transformation, mutagenesis, gene tagging, and phenotype analysis. The effectiveness of these approaches depends on intrinsic biological characteristics such as genome size, ploidy level, reproductive strategy, and genetic redundancy.
A successful model organism must therefore combine both robust physical genomics infrastructure and strong biological genomics capabilities. Additionally, practical traits such as compact growth habit, short generation time, and adaptability to laboratory conditions are essential for enabling high-throughput experimental workflows.

The Need for Grass-Specific Model Systems
Grasses (Poaceae) include the world’s most important crops, such as wheat, rice, maize, and barley. These species are central to global food security, animal feed, and bioenergy production. Consequently, there is a critical need for model systems that accurately represent grass biology.
Initially, researchers attempted to use Arabidopsis as a reference genome for grasses. However, studies revealed limited genomic collinearity between dicots and monocots, reducing its usefulness for identifying genes in cereals. This highlighted the necessity for grass-specific model organisms.
Such models must capture key traits relevant to cereal crops, including:
- Grain development and yield
- Stress tolerance (drought, cold, pathogens)
- Perennial growth and biomass production
- Cell wall composition for bioenergy applications
Rice as a Model and Its Limitations
Rice (Oryza sativa) has been widely adopted as a model for cereal genomics due to its relatively small genome (~441 Mbp) and global importance as a staple food crop. Extensive international efforts have produced genetic maps, expressed sequence tag (EST) collections, and a complete genome sequence.
Rice has contributed significantly to understanding gene function in grasses and has served as a reference for comparative genomics. The concept of a “consensus grass genome” anchored to rice has facilitated gene mapping in species with larger genomes.
However, rice also presents several limitations:
- It is a tropical, semi-aquatic species, making it less suitable for studying temperate grasses
- It has a relatively long life cycle and demanding growth conditions
- Transformation and mutagenesis systems are not universally accessible
- Certain traits relevant to temperate cereals (e.g., cold tolerance, vernalization) are absent
Additionally, genomic rearrangements and lack of microsynteny complicate direct gene transfer between rice and other cereals.
Evolutionary and Genomic Advantages of Brachypodium
Phylogenetically, Brachypodium occupies a strategic position within the grass family. It diverged just before the radiation of major temperate cereal lineages, including wheat and barley. This makes it particularly valuable for comparative genomics within the Pooideae.
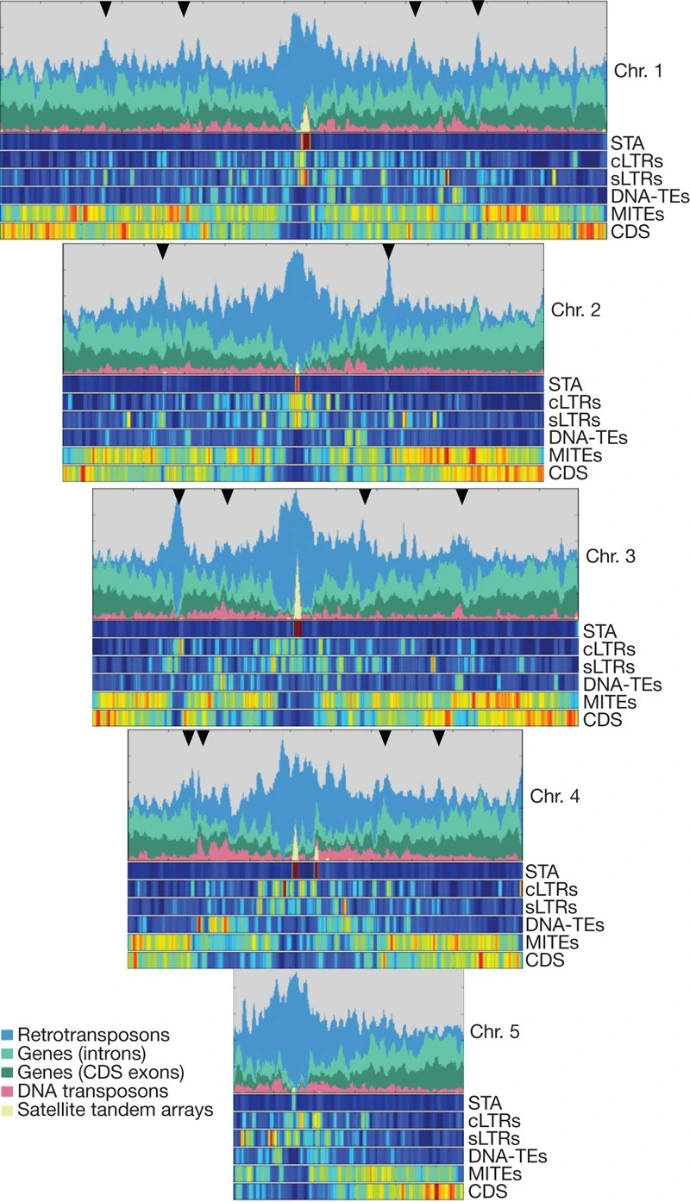
Its genome is characterized by:
- Low repetitive DNA content (<15%)
- Simple chromosomal organization
- Small chromosome number (commonly 2n = 10)
- High recombination frequency
These features facilitate genetic mapping, genome sequencing, and functional studies. Furthermore, the relatively compact genome allows for efficient construction of genomic libraries and rapid identification of genes.
Suitability for High-Throughput Genetics and Transformation
One of the defining strengths of Brachypodium is its compatibility with modern genetic tools, including:
- Tissue culture and regeneration from immature embryos
- Efficient transformation via Agrobacterium or biolistic methods
- Mutagenesis using chemical, physical, or insertional approaches
- Development of transgenic and mutant populations
Compared to major cereals, Brachypodium offers a much faster and more cost-effective platform for testing gene function. For example, while generating transgenic wheat lines may take several years, similar experiments can be completed in Brachypodium within a few months.
Conclusion
In conclusion, Brachypodium distachyon represents a highly efficient and versatile model system for functional genomics in grasses. While rice remains an important reference genome, Brachypodium complements it by providing a system better suited for studying temperate cereals and forage grasses.
Its combination of genomic simplicity, experimental flexibility, and biological relevance makes it an essential tool for modern plant science. As genomic technologies continue to evolve, Brachypodium is expected to play an increasingly important role in bridging the gap between basic research and practical applications in agriculture, bioenergy, and environmental sustainability.